Scientific convergence of immunology and neuroscience
MANY DISEASES, ONE COMMON CAUSE: CHRONIC INFLAMMATION
There is established evidence that chronic inflammation plays a role in causing and advancing many autoimmune diseases including rheumatoid arthritis (RA), inflammatory bowel disease (IBD), psoriasis, and multiple sclerosis. The immune system plays a critical role in fighting infection and maintaining a balanced state in inflammatory pathways.
USING THE BODY’S OWN HEALING REFLEX
In early 2000s, SetPoint Medical’s co-founder, Dr. Kevin Tracey discovered a new biological pathway – the Inflammatory Reflex – that created the possibility of treating chronic autoimmune disease using neurostimulation. The Inflammatory Reflex signals along the vagus nerve to maintain homeostasis in the immune system and maintain a healthy immunologic setpoint.
THE CENTRAL ROLE OF THE VAGUS NERVE
The vagus nerve is the longest nerve in the body and innervates many organs. It is composed of thousands of sensory and motor neurons. The sensory neurons detect and respond to inflammatory mediators by sending signals to the brainstem, where a signal reflexively originates. This new signal is transmitted by the motor neurons within the vagus nerve to the splenic nerve causing the release of norepinephrine in the spleen. Specialized CD4+ T lymphocytes (T-cells) in the spleen, expressing adrenaline receptors, take up the norepinephrine and release acetylcholine, which in turn binds to the α7 nicotinic receptors on immune cells. This reduces the activity of several key intracellular inflammation pathways. The vagus nerve thus plays a central role in dialing down production of inflammatory cytokines.
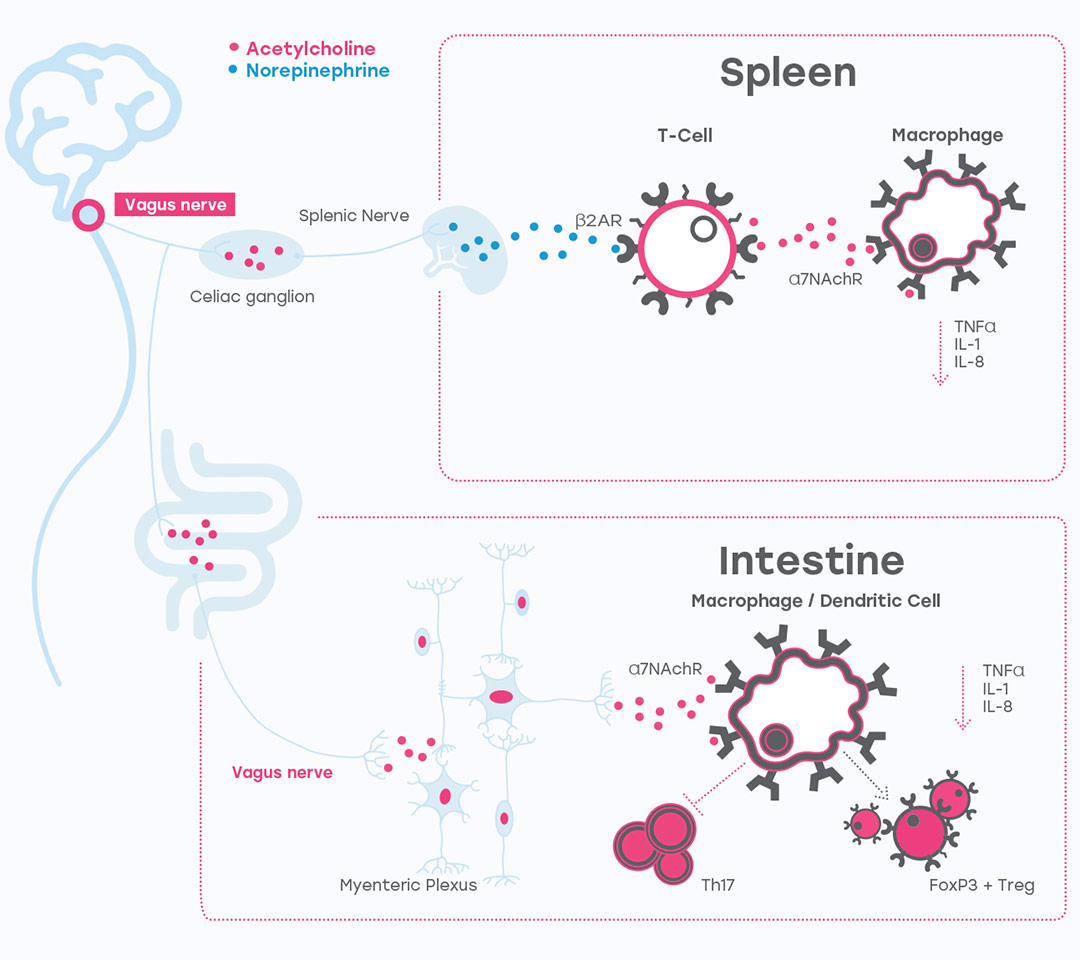
MICROSTIMULATION AT PRECISELY THE RIGHT LOCATION
With the discovery of the Inflammatory Reflex, SetPoint Medical’s researchers set out to characterize this cholinergic anti-inflammatory pathway and harness it for therapeutic use. We identified the specific fibers in the vagus nerve and defined the precise stimulation parameters to activate the Inflammatory Reflex.
Biomarker analysis indicated that the anti-inflammatory response through a single vagus nerve stimulation lasted more than 24 hours. More importantly, this approach suppressed inflammatory cytokines by 30-70% without eliminating their bioavailability, thereby reducing inflammation with the potential to be less immunosuppressive. (Genovese MC et. al, Lancet Rheum, 2020, 2(9):E527-E538)
CAUTION – Investigational device. Limited by United States law to investigational use.